LIFE NEEDS PHOSPHORUS

At the left, a corn leaf's purple blotches indicate phosphorus deficiency. Cells in the blade are losing their ability to photosynthesize and are dying because the corn plant doesn't have enough phosphorus in its body.
In fact, nearly all life on Earth absolutely, utterly, unconditionally needs phosphorus. The word "nearly" is used because in 2010, in arsenic-rich mud a bacterium species was discovered which had evolved to use arsenic instead of phosphorus, arsenic being very similar chemically to phosphorous. Except for that, for many years it's been one of the truisms of science that life simply isn't possible without six "essential elements," one of which is phosphorus.
The six essential elements are: CARBON, HYDROGEN, NITROGEN, OXYGEN, PHOSPHORUS & SULFUR, sometimes memorably known as "CHNOPS".
AT THE MOLECULAR LEVEL
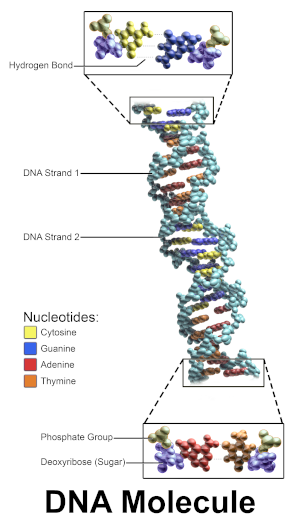 DNA molecule; note phosphate group identified at bottom; image courtesy of Bruce Blaus, via Blausen.com staff (2014). Medical gallery of Blausen Medical 2014. WikiJournal of Medicine 1 (2). DOI:10.15347/wjm/2014.010. ISSN 2002-4436
DNA molecule; note phosphate group identified at bottom; image courtesy of Bruce Blaus, via Blausen.com staff (2014). Medical gallery of Blausen Medical 2014. WikiJournal of Medicine 1 (2). DOI:10.15347/wjm/2014.010. ISSN 2002-4436The reasons phosphorus are necessary are technical and really the backyard naturalist doesn't need to understand them. However, it's good to have access to some of the reasons just in case you want to "look them up." Here are some of the reasons that phosphorus is absolutely necessary for all life (except for that bacterium in arsenic-rich mud and maybe a few other undiscovered freakish beings):
- The DNA and RNA molecules require phosphorus atoms as part of their basic molecular structure (the bacterium used arsenic atoms in its DNA instead)
- In the intracellular energy transfer system, the energy-carrying molecule ATP requires a phosphorus atom (the P in ATP is for phosphorus)
- GTP, the guanosine triphosphate molecule, is similarly used in the synthesis of protein and in "gluconeogenesis"; (the P in GTP also is for phosphorus)
- Molecules known as phospholipids constitute the main structural elements of all cellular membranes (note the "phospho" in "phospholipid")
- Phosphorus, as the second most abundant mineral in the human body, makes up about 1% of body weight. Most of the phosphorus is in bones and teeth. Phosphate constitutes part of the general bone matrix.
IS THE EARTH'S PHOSPHORUS DISAPPEARING?
oxygen......46.1%
silicon.....28.2%
aluminum....8.23%
calcium.....5.63%
sodium......4.15%
magnesium...2.36%
potassium...2.33%
titanium....2.09%
hydrogen....0.57%
phosphorus..0.14%
Phosphorus is the 11th most common element on Earth, so there's not much of a chance that earthlings will ever run out of it. However, if we ask which of the absolutely necessary elements we need to worry about becoming unavailable in a form usable for Life on Earth, it turns out that the answer is phosphorus.
In fact, Isaac Asimov, an important early science writer, described phosphorus as "life's bottleneck." Asimov noticed that certain mineral elements are more common in organism bodies than in the environment in which those bodies lived. For that to be so, organisms must be concentrating that element in themselves, which is no simple matter.
Asimov noted that phosphorus composes about 0.12% of an average soil, yet a much greater percentage of an alfalfa plant's body, about 0.7%, is phosphorus. Therefore, the "concentration factor" for phosphorus in alfalfa is about 5.8 (0.7/0.12).
No other mineral element even comes close to having a concentration factor as great as phosphorus's. The closest is sulfur with 2.0, then chlorine with 1.5. All the rest have less than a factor of 1. Clearly, to that alfalfa plant, phosphorus was important.
Therefore, if on Earth or any ecosystem there are more and more organisms needing mineral elements, and/or if the living ecosystem is more and more depleted of its resources, which absolutely necessary mineral element will come into short supply first, unless you're that arsenic-crazed bacterium in the mud?
Phosphorus.
The first words in a 2020 paper by Christine Alewell and others, available at the prestigious Nature.Com website and entitled "Global phosphorus shortage will be aggravated by soil erosion" flatly states: "Soil phosphorus (P) loss from agricultural systems will limit food and feed production in the future."
PHOSPHORUS & EUTROPHICATION
 Algal bloom in a creek whith eutrophication exacerbated by drought and low discharge; image courtesy of John Robert McPherson in Australia & Wikimedia Commons
Algal bloom in a creek whith eutrophication exacerbated by drought and low discharge; image courtesy of John Robert McPherson in Australia & Wikimedia CommonsA problem is that phosphorus dissolves in water and leaches from our soils, is eroded away as soil particles, is removed from the land in the crops we harvest, and flows down our drains whenever we use phosphorus-rich detergents or flush the commode. Not only does this mean that the Earth's human-disturbed soils are constantly losing phosphorus, but also in waterways, lakes and the ocean, phosphorous -- because it's so important as a nutrient -- acts as fertilizer, causing problems such as the algal bloom at the right. That creek is suffering from eutrophication.
Eutrophication occurs when too many nutrients are added to a body of water -- especially nitrates and phosphates -- frequently due to runoff from the land and sewage discharge, causing dense growth of plant life.
This dense, sometimes explosive growth of plant life, such as during an algal bloom, can result in large populations of animals dying from lack of oxygen. As more algae and plants grow, other die. Bacteria decomposing the dead organisms use up the water's oxygen. Also, algae that produce oxygen during the day may consume oxygen at night.
Thus, when phosphorus escapes from the soil, it causes deficiency problems in the soil, then in water downstream, problems by being tobecause not so much of it is needed.
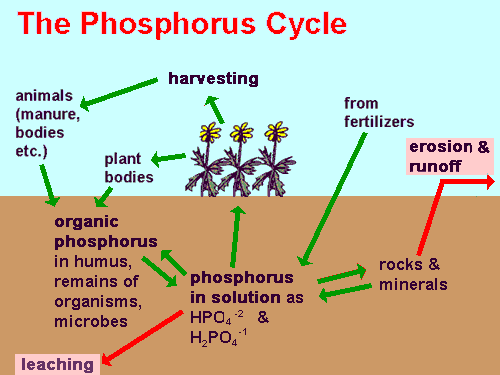
Here's a generalized "phosphorus cycle," with the two red boxes showing where phosphorus is lost from the ecosystem -- where it begins its journey to the ocean.